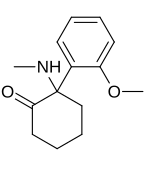
Methoxyketamine
Names
IUPAC name
2-(2-Methoxyphenyl)-2-(methylamino)cyclohexanone
Identifiers
7063-51-6 Y 6728-62-7 (HCl ) Y
ChemSpider
UNII
U4I8JIS24N Y 2OVB5UO35R (HCl ) Y
InChI=1S/C14H19NO2/c1-15-14(10-6-5-9-13(14)16)11-7-3-4-8-12(11)17-2/h3-4,7-8,15H,5-6,9-10H2,1-2H3
Key: OYAUVHORXFUVAJ-UHFFFAOYSA-N
InChI=1/C14H19NO2/c1-15-14(10-6-5-9-13(14)16)11-7-3-4-8-12(11)17-2/h3-4,7-8,15H,5-6,9-10H2,1-2H3
Key: OYAUVHORXFUVAJ-UHFFFAOYAM
Properties
C 14 H 19 N O 2
Molar mass
−1
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
Chemical compound
Methoxyketamine or 2-MeO-2-deschloroketamine is a designer drug of the arylcyclohexylamine class first reported in 1963.[1] It is an analog of ketamine in which the chlorine atom has been replaced with a methoxy group . Its synthesis by rearrangement of an amino ketone has been reported.[2] As an arylcyclohexylamine, methoxyketamine most likely functions as an NMDA receptor antagonist . It produces sedative, hallucinogenic, and (at high doses) anesthetic effects, but with a lower potency than ketamine itself.
See also References
^ BE 634208, Stevens, Calvin L., "Amino ketones", published 1963 ^ Stevens, Calvin L.; Thuillier, Andre; Taylor, K. Grant; Daniher, Francis A.; Dickerson, James P.; Hanson, Harry T.; Nielsen, Norman A.; Tikotkar, N. A.; Weier, Richard M. (1966). "Amino Ketone Rearrangements. VII.1 Synthesis of 2-Methylamino-2-Substituted Phenylcyclohexanones". The Journal of Organic Chemistry . 31 (8): 2601. doi :10.1021/jo01346a034.
Psychedelics (5-HT2A
Benzofurans Lyserg‐ Phenethyl‐
2C-x 25x -NBx
25x -NB 25x -NB3OMe
25B-NB3OMe
25C-NB3OMe 25D-NB3OMe
25E-NB3OMe
25H-NB3OMe
25I-NB3OMe 25N-NB3OMe
25P-NB3OMe
25T2-NB3OMe
25T4-NB3OMe
25T7-NB3OMe
25TFM-NB3OMe 25x -NB4OMe
25B-NB4OMe
25C-NB4OMe 25D-NB4OMe
25E-NB4OMe
25H-NB4OMe
25I-NB4OMe 25N-NB4OMe
25P-NB4OMe
25T2-NB4OMe
25T4-NB4OMe
25T7-NB4OMe
25TFM-NB4OMe 25x -NBF 25x -NBMD
25B-NBMD
25C-NBMD
25D-NBMD
25E-NBMD
25F-NBMD
25H-NBMD
25I-NBMD 25P-NBMD
25T2-NBMD
25T7-NBMD
25TFM-NBMD 25x -NBOH 25x -NBOMe Atypical structures
25x -NMx
25B-NMe7BF
25B-NMe7BT
25B-NMe7Bim
25B-NMe7Box
25B-NMe7DHBF
25B-NMe7Ind
25B-NMe7Indz
25B-NMePyr
25I-NMe7DHBF
25I-NMeFur
25I-NMeTHF
25I-NMeTh N-(2C)-fentanyl
N-(2C-B) fentanyl
N-(2C-C) fentanyl
N-(2C-D) fentanyl
N-(2C-E) fentanyl
N-(2C-G) fentanyl
N-(2C-H) fentanyl
N-(2C-I) fentanyl
N-(2C-IP) fentanyl
N-(2C-N) fentanyl
N-(2C-P) fentanyl
N-(2C-T) fentanyl
N-(2C-T-2) fentanyl
N-(2C-T-4) fentanyl
N-(2C-T-7) fentanyl
N-(2C-TFM) fentanyl
3C-x 4C-x DOx HOT-x MDxx Mescaline (subst.) TMAs
TMA
TMA-2
TMA-3
TMA-4
TMA-5
TMA-6 Others
Piperazines Tryptamines
alpha -alkyltryptaminesx -DALT x -DET x -DiPT x -DMT
4,5-DHP-DMT 2,N,N-TMT 4-AcO-DMT 4-HO-5-MeO-DMT 4,N,N-TMT
4-Propionyloxy-DMT 5,6-diBr-DMT
5-AcO-DMT 5-Bromo-DMT 5-MeO-2,N,N-TMT
5-MeO-4,N,N-TMT
5-MeO-α,N,N-TMT
5-MeO-DMT 5-N ,N -TMT 7,N,N-TMT α,N,N-TMT (Bufotenin) 5-HO-DMT DMT Norbaeocystin (Psilocin) 4-HO-DMT (Psilocybin) 4-PO-DMT x -DPT Ibogaine-related x -MET x -MiPT Others
Others
Dissociatives (NMDAR antagonists )
Deliriants (mAChR antagonists ) Others
AMPAR Tooltip α-Amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor KAR Tooltip Kainate receptor NMDAR Tooltip N-Methyl-D-aspartate receptor
Group I
mGluR1 Tooltip Metabotropic glutamate receptor 1 mGluR5 Tooltip Metabotropic glutamate receptor 5
Group II
mGluR2 Tooltip Metabotropic glutamate receptor 2 mGluR3 Tooltip Metabotropic glutamate receptor 3
Group III
mGluR4 Tooltip Metabotropic glutamate receptor 4
Antagonists: CPPGMAP4
MPPG
MSOP
MTPG
UBP-1112 mGluR6 Tooltip Metabotropic glutamate receptor 6
Antagonists: CPPGMAP4
MPPG
MSOP
MTPG
UBP-1112 mGluR7 Tooltip Metabotropic glutamate receptor 7
Antagonists: CPPGMAP4
MMPIP MPPG
MSOP
MTPG
UBP-1112
XAP044 ; Negative allosteric modulators: ADX71743 mGluR8 Tooltip Metabotropic glutamate receptor 8
Antagonists: CPPGMAP4
MPPG
MSOP
MTPG
UBP-1112