Ayanin
Names
IUPAC name
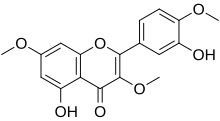
3′,5-Dihydroxy-3,4′,7-trimethoxyflavone
Systematic IUPAC name
5-Hydroxy-2-(3-hydroxy-4-methoxyphenyl)-3,7-dimethoxy-4H -1-benzopyran-4-one
Other names
3,7,4'-Tri-O -methylquercetin 3,7,4'-Trimethylquercetin
Identifiers
572-32-7 Y
Interactive image Interactive image
ChEBI
CHEBI:27825 Y
ChEMBL
ChEMBL74898 Y
ChemSpider
4444274 Y
UNII
YA465UF3LK Y
InChI=1S/C18H16O7/c1-22-10-7-12(20)15-14(8-10)25-17(18(24-3)16(15)21)9-4-5-13(23-2)11(19)6-9/h4-8,19-20H,1-3H3
Y Key: KPCRYSMUMBNTCK-UHFFFAOYSA-N
Y InChI=1/C18H16O7/c1-22-10-7-12(20)15-14(8-10)25-17(18(24-3)16(15)21)9-4-5-13(23-2)11(19)6-9/h4-8,19-20H,1-3H3
Key: KPCRYSMUMBNTCK-UHFFFAOYAE
COC1=C(C=C(C=C1)C2=C(C(=O)C3=C(C=C(C=C3O2)OC)O)OC)O
O=C1c3c(O/C(=C1/OC)c2ccc(OC)c(O)c2)cc(OC)cc3O
Properties
C 18 H 16 O 7
Molar mass
−1
Density
1.454 g/mL
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
Chemical compound
Ayanin is an O -methylated flavonolO -methylated derivative of quercetin .
It can be found in Croton schiedeanus [1]
Biosynthesis The enzyme 3,7-dimethylquercetin 4'-O-methyltransferase uses S -adenosyl methioninerhamnazin to produce S -adenosylhomocysteine
References
^ Rao, Koppaka V.; Owoyale, Jacob A. (1976). "Partial methylation of quercetin: Direct synthesis of tamarixetin, ombuin and ayanin". Journal of Heterocyclic Chemistry . 13 (6): 1293–1295. doi :10.1002/jhet.5570130629 .
Flavonols and their conjugates
Backbone
Flavonols
Aglycones Conjugates
Glycosides of herbacetin Glycosides of kaempferol
Afzelin (Kaempferol 3-rhamnoside)Astragalin (kaempferol 3-O-glucoside)Kaempferitrin (kaempferol 3,7-dirhamnoside)Juglanin (Kaempferol 3-O-arabinoside)Kaempferol 3-alpha-L-arabinopyranoside
Kaempferol 3-alpha-D-arabinopyranoside
Kaempferol 7-alpha-L-arabinoside
Kaempferol 7-O-glucoside Kaempferol 3-lathyroside
Kaempferol 4'-rhamnoside
Kaempferol 5-rhamnoside
Kaempferol 7-rhamnoside
Kaempferol 7-O-alpha-L-rhamnofuranoside
Kaempferol 3-xyloside
Kaempferol 7-xyloside
Robinin (kaempferol-3-O-robinoside-7-O-rhamnoside)Kaempferol 3-O-rutinoside Sophoraflavonoloside (Kaempferol 3-O-sophoroside)
Trifolin (Kaempferol 3-O-beta-D-galactoside) Glycosides of myricetin Conjugates of quercetin
O -Methylated flavonols
Aglycones Glycosides
of isorhamnetin
Narcissin (Isorhamnetin 3-O-rutinoside)
Isorhamnetin 3-O-glucoside
Tamarixetin 7-rutinoside other
Azalein (Azaleatin 3-O-α-L-rhamnoside)Centaurein (Centaureidin 7-O-glucoside)
Eupalin (Eupalitin 3-0-rhamnoside)Eupatolin (Eupatolitin 3-O-rhamnoside)Jacein (Jaceidin 7-O-glucoside)
Patulitrin (Patuletin 7-O-glucoside
Xanthorhamnin (Rhamnetin glycoside)
Derivative flavonols
Aglycones
Noricaritin
Dihydronoricaritin Glycosides
Pyranoflavonols
Furanoflavonols
Semisynthetic